MADCO-Accelerated Multidimensional Diffusion MRI
Although multidimensional diffusion/relaxation NMR experiments are widely used in materials sciences and engineering applications, preclinical and clinical MRI applications of these techniques were not feasible. Moreover, higher-field MRI scanners posed another obstacle to translation of this NMR method. Their specific absorption rate (SAR) limits the use of multi-echo or CPMG pulse trains, so that the large amounts of data required by these methods cannot be collected in vivo due to exceedingly long scan times. Therefore, the primary challenges this invention overcomes are the migration of NMR methods to MRI and to vastly shorten scan times.
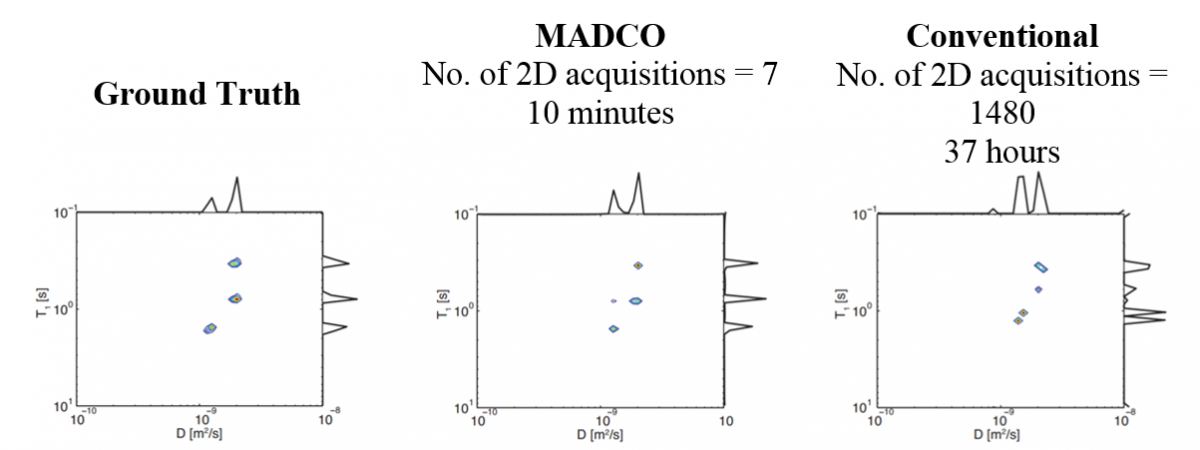
The “Marginal Distributions Constrained Optimization (MADCO)” methodology provides a novel framework to accelerate and improve the reconstruction of multidimensional NMR relaxation/diffusion spectra suitable for use in MRI applications on a voxel-by-voxel basis. This approach uses 1D acquired spectra as a priori information to estimate a 2D (or higher dimensional) spectrum. 1D marginal distributions are used as constraints when the 2D spectra are reconstructed.
The MADCO methodology was demonstrated with polyvinylpyrrolidone-water solution phantoms, which contains 3 species of diffusivities and T1 relaxation times (i.e., 3 peaks in the D-T1 space). With a reasonably accurate estimate of the 1D marginal distributions, MADCO accelerated the acquisition of the 2D spectra by more than a factor of 200 (i.e., two orders of magnitude) with a high level of accuracy, compared to conventional, unconstrained methods.
Competitive Advantages:
- Disruptive technologies can positively impact market penetration in a large and growing medical sector. In 2016, an estimated 39 million magnetic resonance imaging (MRI) procedures were performed – reflecting an average annual growth rate of ~4% since 2011. In the same year, over 8500 clinical sites offered the procedure
- Metabolomics; increased use in quantitative and metabolic screening applications, where the bottleneck in exploiting the higher information content of multidimensional spectra is the time required to acquire the magnetic resonance data
- The MADCO method would accelerate and improve the reconstruction of multidimensional NMR relaxation/diffusion spectra suitable for use in MRI, where reducing the amount of data required, reduces the time of scanning, and reduces the cost
- Imaging procedures such as MRIs use lower radiation levels compared with CT and PET scans, lowering patients risk
- Shortens scan time using MADCO, resulting in reduced patient discomfort as well as improve diagnostic accuracy
- Decreases probability of overuse and increases patient benefit for what can be a costly diagnostic procedure
Commercial Applications:
- Incorporation into neuro or whole-body MRI suites to measure changes in neural and other human tissue within a clinically feasible time-frame
- Monitoring and mapping brain injury and other pathologies, including inflammation
- Cardiovascular disease; e.g., stroke, thrombolysis
- Dental diseases for which standard x-rays do not provide adequate resolution
- New quantitative imaging biomarkers for improved radiological assessment of normal and abnormal development, various diseases and disorders that have neuroanatomical and microstructural sequelae (e.g., TIA, cancer, Alzheimer’s disease, inflammatory diseases)
- Investigation of fixed tissue, living organotypic neural or other tissue or pre-clinical in vivo animal models in a feasible time frame
- Producing new quantitative radiological biomarkers, stains or contrasts. Providing a new method for scanning and assessing foods safety and storage life
- Characterization of rock and soil samples in situ or in a laboratory setting for the oil, gas and water exploration fields
- Assessing materials and material properties, such as polymers, gels, composites, concrete, and other man-made and natural materials
Patents
- US
Provisional (PRV) 62/373,497
Filed on 2016-08-11
Status: Abandoned - Patent Cooperation Treaty
(PCT) PCT/US2017/046615
Filed on 2017-08-11
Status: Expired - Australia
National Stage 2017310513
Filed on 2019-01-23
Status: Issued - Canada
National Stage 3032154
Filed on 2017-08-11
Status: Pending - European Patent
National Stage 17755012.6
Filed on 2019-02-05
Status: Issued - Japan
National Stage 2019-506625
Filed on 2019-02-07
Status: Issued - US Patent 11,415,652
Filed on 2019-02-08
Status: Issued - US Patent 11,846,690
Filed on 2022-07-13
Status: Issued
Publications
- Benjamini D, et al. Clinically feasible relaxation-diffusion correlation MRI using MADCO.
- Benjamini D, et al. Towards clinically feasible relaxation-diffusion correlation MRI using MADCO.
- Bai R, et al. Fast, accurate 2D-MR relaxation exchange spectroscopy (REXSY): Beyond compressed sensing.
- Benjamini D, et al. Imaging Local Diffusive Dynamics Using Diffusion Exchange Spectroscopy MRI.
- Benjamini D, et al. Water exchange in a white matter tissue phantom measured using clinically feasible diffusion exchange spectroscopy (DEXSY) MRI.
- Benjamini D, et al. Use of marginal distributions constrained optimization (MADCO) for accelerated 2D MRI relaxometry and diffusometry.
- Benjamini D, et al. Marginal distributions constrained optimization (MADCO) used to accelerate 2D MRI relaxometry.
Collaborations
- Materials Available
- Licensing
- Collaboration