T Cell Receptors Targeting p53 Mutations for Cancer Immunotherapy and Adoptive Cell Therapy
The tumor protein p53 is a cell cycle regulator. It responds to DNA damage by triggering the DNA repair pathway and allowing cell division to occur or inducing cell growth arrest, cellular senescence, and/or apoptosis. p53 therefore acts as a tumor suppressor by preventing uncontrolled cell division. However, mutations in p53 that impair its cell cycle regulatory functions can induce uncontrolled cell division leading to cancer. Certain p53 mutations, termed ‘hotspot’ mutations, occur at high frequency across patients and diverse cancer types, such as cholangiocarcinoma, melanoma, colon cancer, rectal cancer, ovarian cancer, endometrial cancer, non-small cell lung cancer (NSCLC), glioblastoma, uterine cervical cancer, head and neck cancers, breast cancer, pancreatic cancer, and bladder cancer. Novel therapeutics that specifically target mutant p53 proteins may be useful for the treatment of many common malignancies.
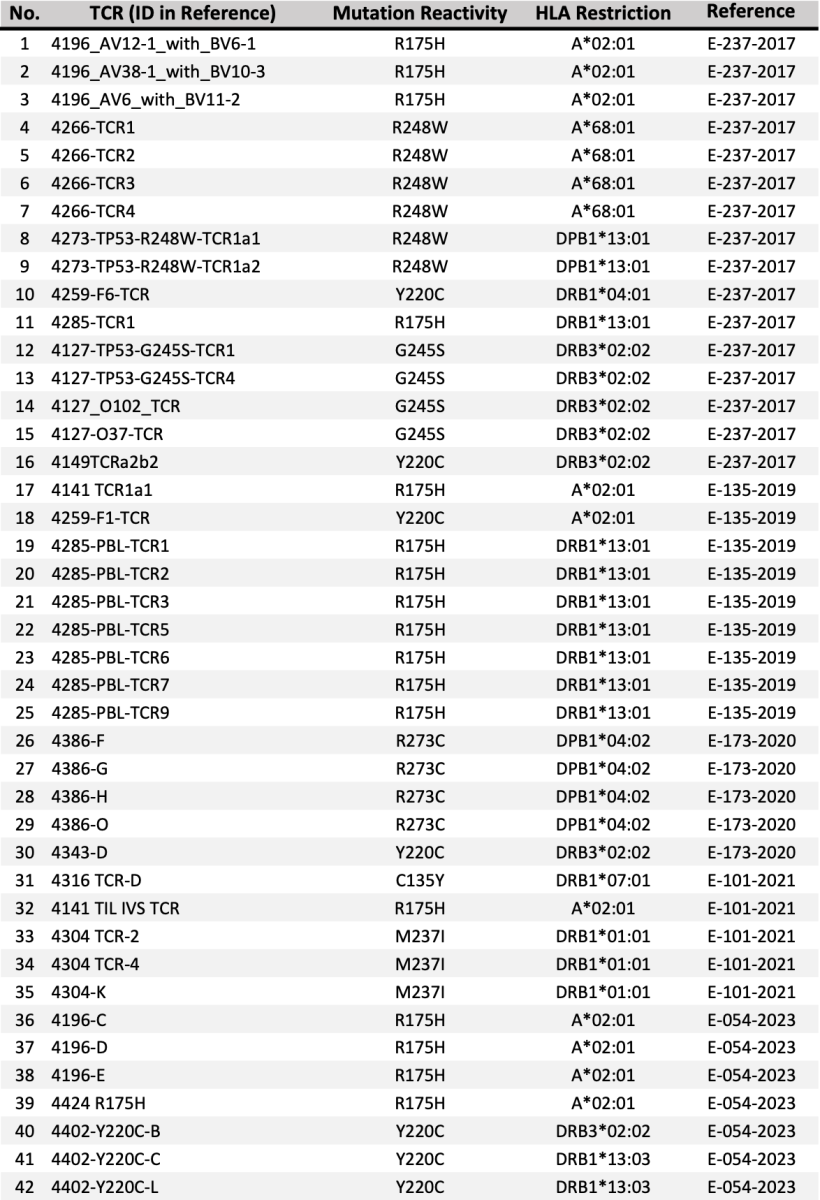
Researchers at the National Cancer Institute (NCI) have identified a collection of novel T-cell receptors (TCRs) targeting defined hotspot mutations in the p53 tumor suppressor protein (Table 1). These p53 hotspot mutations, including I135Y, R175H, Y220C, M237I, G245D, G245S, R248L, R248Q, R248W, R249S, R273C, R273L, R273H and R282W, are prevalent in cancer cells and are therefore attractive targets for TCR T cell therapy. These TCRs are expected to specifically eliminate human cancer cells bearing these p53 mutations upon adoptive transfer into cancer patients.
The NCI Surgery Branch is seeking research co-development partners and/or licensees for these T Cell Receptors Targeting Mutated p53.

Competitive Advantages:
- Mutated p53 is not expressed in normal cells, suggesting the TCR therapy will have a low toxicity profile
- Variety of HLA-restriction elements: extends the applicability of TCRs as they recognize mutated p53 variants in the context of multiple HLA molecules
Commercial Applications:
- T-cell therapy against a variety of human cancers as p53 is a commonly mutated gene in several human cancers including melanoma, breast cancer, colon cancer, and bladder cancer
- TCRs can be used in diagnostic tools to identify presence of p53 hotspot mutations in cancer cells
- Use of the TCRs in chimeric proteins for research purposes in cancers with mutated p53
Patents
- US Patent 11,939,365
Filed on 2020-03-26
Status: Issued - US
Provisional (PRV) 62/565,383
Filed on 2017-09-29
Status: Abandoned - US
Provisional (PRV) 62/565,464
Filed on 2017-09-29
Status: Abandoned - Patent Cooperation Treaty
(PCT) PCT/US2018/051280
Filed on 2018-09-17
Status: Expired - Australia
National Stage 2018342245
Filed on 2018-09-17
Status: Issued - Canada
National Stage 3080274
Filed on 2018-09-17
Status: Pending - China
National Stage 201880063656.4
Filed on 2018-09-17
Status: Pending - European Patent
National Stage 18782605.2
Filed on 2018-09-17
Status: Issued - Israel
National Stage 273516
Filed on 2020-03-23
Status: Issued - Japan
National Stage 2020-517553
Filed on 2020-03-26
Status: Issued - South Korea
National Stage 10-2020-7012343
Filed on 2020-04-28
Status: Issued - Singapore
National Stage 11202002635R
Filed on 2018-09-17
Status: Abandoned - US
National Stage 16/650,696
Filed on 2020-03-25
Status: Pending - Hong Kong
European patent (EP) 62020021274.9
Filed on 2020-11-30
Status: Issued - Patent Cooperation Treaty
Patent Cooperation Treaty Combined (PCT COMB) PCT/US2018/051285
Filed on 2018-09-17
Status: Expired - Australia
National Stage 2018342246
Filed on 2018-09-17
Status: Issued - Brazil
National Stage BR112020006012-7
Filed on 2018-09-17
Status: Pending - Canada
National Stage 3077024
Filed on 2018-09-17
Status: Pending - China
National Stage 201880074539.8
Filed on 2018-09-17
Status: Issued - Costa Rica
National Stage 2020-0170
Filed on 2018-09-17
Status: Pending - Eurasian Patent
National Stage 202090757
Filed on 2018-09-17
Status: Issued - European Patent
National Stage 18780006.5
Filed on 2018-09-17
Status: Issued - Israel
National Stage 273515
Filed on 2020-03-23
Status: Issued - India
National Stage 202047013911
Filed on 2018-09-17
Status: Pending - Japan
National Stage 2020-517556
Filed on 2018-09-17
Status: Issued - South Korea
National Stage 10-2020-7012344
Filed on 2020-04-28
Status: Issued - Mexico
National Stage MX/a/2020/003504
Filed on 2018-09-17
Status: Issued - New Zealand
National Stage 763023
Filed on 2020-03-27
Status: Pending - Singapore
National Stage 11202002636P
Filed on 2018-09-17
Status: Pending - Hong Kong
European patent (EP) 62020021272.3
Filed on 2020-11-30
Status: Issued - Brazil
Divisional (DIV) BR122021018454-2
Filed on 2021-09-16
Status: Pending - Japan
Divisional (DIV) 2023-123893
Filed on 2023-07-28
Status: Issued
Publications
- Malekzadeh P, et al. Antigen experienced T cells from peripheral blood recognize p53 neoantigens.
- Deniger et al. T-cell responses to TP53 “hotspot” mutations and unique neoantigens expressed by human ovarian cancers.
- Malekzadeh P, et al. Neoantigen screening identified broad TP53 mutant immunogenicity in patients with epitheleal cancers.
Collaborations
- Licensing
- Collaboration